Valence electrons are electrons in the highest energy level of an atom, usually at the outer level. These electrons take part in a bonding process. All neutral atoms have few electrons. Neutral atoms always have the same number of electrons and protons (also known as the atomic number).
Valence electrons contribute more heavily than valency, while all electrons contribute to some extent. The combination of valence electrons from two or more atoms can form chemical bonds between atoms. The sharing or donation of valency between atoms is necessary for the formation of chemical bonds.
What is Valency?
The valence of an element is the total number of valence electrons present in the element. A chemical compound is a mixture of two or more elements in a specific ratio. A stable compound is when one atom of one element combines with several atoms of another element. The binding capacity of these ingredients varies.
Significance of Valence Electrons
- The reactivity of an atom is directly related to the number of valency.
- Electronegativity is influenced by the valency of an atom.
- The affinity of an atom for an electron is determined by its valence electron.
- Ionization energy is most likely due to the valency of an atom.
- Effect of valency in a stable atom.
- The valency of an atom control its chemical and physical properties.
- For maximum stability, each valence shell of an atom must be filled with 8 electrons. Hydrogen and helium defy this trend by invariably being left with only two electrons in their outer shell.
- Electrons in a closed shell (inner shell) remain close to the nucleus and cannot escape. The outer (valence) electrons can participate in bonding by attaching to or transferring to another atom.
Characteristics of Valence Electrons?
- valency are found in the outer shell of electrons. However, the inner layer of transition metal allows this to happen.
- Atoms with a full set of electrons (octet or doublet) have no chemical activity.
- They absorb or emit energy in the form of small packets of energy (photons).
- valency are used to refer to elements such as metals, nonmetals, or metalloids, depending on their electrical conductivity.
Electron Dot Diagrams?
Due to the importance of valence electrons, atoms are represented in simple diagrams using only valency. These are called electronic dot diagrams and three are shown below. In this type of diagram, the element’s chemical symbol is surrounded by dots representing valency. Draw the points as a rectangle around the signal element, with a maximum of two points on each side. An element never has more than eight valency, so an atom cannot have more than eight valence electrons.
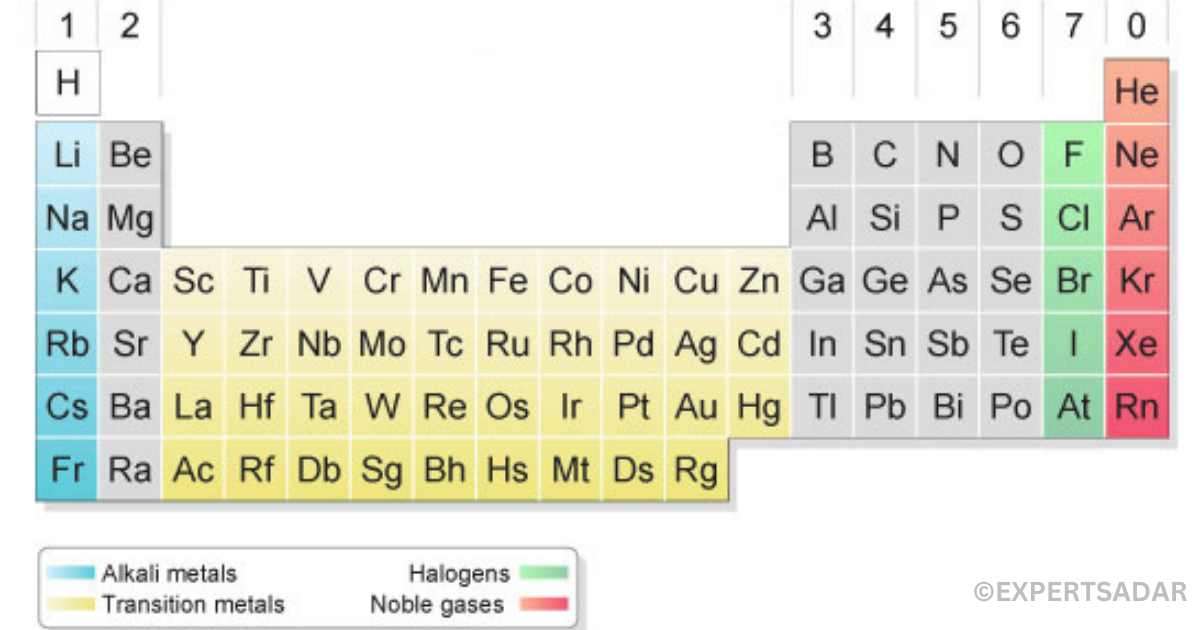
Valence electrons in the periodic table?
The periodic table of elements shows the number of valency in an atom (refer to the periodic table in the figure below). In each row or periodic table, the number of valency in groups 1-2 and 13-18 increases from one element to another. Each element in each column or group of the table has the same valency. This explains why all compounds in a group have the same chemical properties.
You can determine the number of valency in group 1-2 and 13-18 elements directly from the periodic table. The simple periodic table below shows this. It shows the number of valency in each group. It is very difficult to determine the number of valency for groups 3-12 groups.
Valence electrons in chemical reactions?
The number of electrons in the outer valence shell of an atom determines its bonding. The periodic table of elements groups elements with the same number of valence electrons together. In general, the fewer electrons an atom has in its valence shell, the more active it is. Therefore, group 1 alkali metals are very reactive, and lithium, sodium, and potassium are the most reactive of all the metals.
Each atom in the valence layer is more stable or more reactive. An atom can share electrons with neighboring atoms (covalent bonds) or accept electrons from other atoms (ionic bonds). Another type of ionic bond involves an atom donating some of its electrons to another atom. It also works because you can get a full waltz by leaving the entire outer layer. The transfer of electrons bonds the two atoms together. A chemical bond binds atoms together, forming molecules or ionic compounds. Five categories can categorize relationships.
- Ionic bond
- Covalent bonds
- Dative covalent bonds, sometimes referred to as coordinate covalent bonds.
- Hydrogen bonds
- Steel belts
Valence electrons are responsible for determining the electrical conductivity of an element.
Category: Chemical Bonds | electronics
Blog By:- ExpertSadar